
Overview
Shane Crotty, Ph.D., and his team study immunity against infectious diseases. They investigate how the immune system remembers infections and vaccines. By remembering infections and vaccines, the body is protected from becoming infected in the future. Vaccines are one of the most cost-effective medical treatments in modern civilization and are responsible for saving millions of lives. Yet, good vaccines are very difficult to design, and very few new vaccines have been made in the past 10 years. A better understanding of immune memory will facilitate the ability to make new vaccines. Dr. Tony Fauci, NIH, referred to some of the Crotty lab work as “exceedingly important to the field of immunogen design.”
COVID-19 Immunology
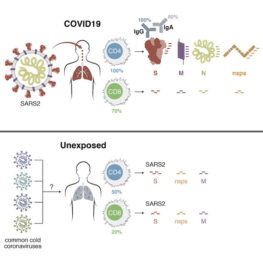
Credit: Cell, May 2020
Dr. Crotty is a member of the LJI Coronavirus Task Force. The Crotty Lab, in close collaboration with the lab of LJI Professor Alessandro Sette, Dr.Biol.Sci., was the first to publish a detailed analysis of the immune system’s response to SARS-CoV-2, the virus that causes COVID-19 (Cell, May 2020). The made a number of important findings. Most importantly, it showed that the immune system activates all three major branches of “adaptive immunity” (which learns to recognize specific viruses) to try to fight the virus: CD4 “helper” T cells , CD8 “killer” T cells, and antibodies. The LJI team found good immune responses to multiple different parts of SARS-CoV-2 (imagine the virus is made out of legos, and the immune system can recognize different individual legos), including the Spike protein, which is the main target of almost all COVID-19 vaccine efforts. This research helped dispel fears that the virus would elude efforts to create an effective vaccine. This scientific study had served as an important benchmark of immune responses for clinical studies and COVID-19 vaccine studies around the world, as evidenced by how extensively the study is being quoted (cited) in the scientific literature. It has also become the #1 most public attention getting Cell paper ever, according to Almetric.
Drs. Crotty and Sette also found that crossreactive immune memory appeared to exist in ~50% of unexposed, healthy people (Cell, May 2020). They inferred this might be due to previous infections with common cold coronaviruses. The LJI team went on to show that crossreactive memory T cells that recognize common cold coronaviruses also recognize matching sites on SARS-CoV-2 (Science, August 2020). The research may explain why some people have milder COVID-19 cases than others—though Crotty and Sette emphasize that this is speculation and much more data is needed (Nature Reviews Immunology, July 2020). Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Disease, has shared this research with Congress, describing the study as “work we really need to pursue. We’re just at the cusp of really understanding the importance of this type of response in COVID.”
The Crotty Lab’s COVID-19 research has informed vaccine efforts worldwide. As highlighted by Voice of America, this work builds on what scientists have learned about past vaccines and the experimental approaches that may bring a COVID-19 vaccine to patients quickly. As Dr. Crotty has said, “I think there is good reason for optimism, but it’s not like in the movies. It takes a lot of people and a lot of time and effort to puzzle it out.”
Cells important for vaccines
Most vaccines work because they generate antibodies. Dr. Crotty made a seminal finding in how this process occurs (Science 2009). Dr. Crotty said it has been well established that antibody production is a multi-step process that involves interactions between several cellular players, key among them CD4 “helper” T cells, which are disease-fighting white blood cells that tell other cells to produce antibodies in response to infections. “There were different flavors of these helper T cells and, for many years, the wrong cell type was identified.” Dr. Crotty’s team showed that a new type of helper cell, “Tfh,” are required for antibody responses. His lab team set out to understand the inner workings of these critical cells in protective immune responses. “We discovered that the BCL6 gene was an on and off switch—a master regulator—in this process. In a series of experiments, we showed that if you turn on this gene, you get more helper cells, the Tfh type, and it is those cells that are telling the B cells to produce antibodies,” he said. The laboratory is now internationally recognized as the leader in Tfh cell biology, having elucidated critical aspects of Tfh cellular and molecular biology, which have been found to be not only important for vaccine biology but also in allergies, autoimmune diseases, and cancers.
New vaccine strategies
Dr. Crotty has a major focus studying human immune responses to vaccines. His lab is hard at work on candidate HIV vaccines with the CHAVID consortium. His lab is also hard at work on vaccine strategies for influenza, strep throat, and COVID-19. The Crotty lab studies new vaccine ideas and strategies that may be applicable to many diseases, based on a fundamental understanding of the underlying immune responses, and how the cells of the immune system interact. Last year the Crotty lab published that a simple slow delivery immunization strategy greatly improves the quality of vaccine responses (Cell 2019). The new strategy hinges on immune cell teamwork. Lead author Kimberly Cirelli, Ph.D., said, “It’s like physical training—you start off weak and then keep going back to the gym to get stronger. The germinal center is the gym and the B cells have to repeatedly go back to undergo rounds of selection to get better binding.” In 2020 the Crotty lab collaborated with Darrell Irvine’s lab at MIT to demonstrate a novel vaccine strategy (Nature Medicine 2020), with the labs hard at work on more new vaccine approaches.
Other background
Dr. Crotty regularly does media outreach on vaccines and immunity to infectious diseases. Dr. Crotty is also the author of Ahead of the Curve, a biography of Nobel laureate scientist David Baltimore, published in 2001, and reviewed in The Wall Street Journal and other publications. He earned his B.S. in Biology and Writing from Massachusetts Institute of Technology (MIT) in 1996, and his Ph.D. in Molecular Biology/Virology from the University of California, San Francisco (UCSF) in 2001.
VOA-TEK: COVID-19 Research
Starting at 19:44, take a look back at how the Crotty Lab applied their research to support SARS-CoV2 vaccine development in the early days of the pandemic.
Featured publications
Engineered immunogen binding to alum adjuvant enhances humoral immunity
Lab Members
 SC
SC
Shane Crotty, Ph.D.
Chief Scientific Officer Center for Autoimmunity and Inflammation, Center for Sex-based Differences in the Immune System, Center for Vaccine Innovation - DirectorResearch Projects
Designing Vaccines: Differentiation and Function of Follicular Helper CD4 T Cells (Tfh Cells)
A video lecture on Tfh cell biology can be seen here. Germinal centers are the critical sites for the development
HIV Vaccine Research (AIDS Vaccine Research)
Broadly neutralizing antibodies have been discovered in ~5% of HIV+ individuals. Some of those antibodies (HIV bnAbs) are capable of
From the lab

Ivy Phung works to build better vaccines
How a Ph.D. student found a place in the intense world of HIV vaccine research

La Jolla Institute for Immunology appoints Shane Crotty, PhD, as Chief Scientific Officer
Dr. Crotty will lead LJI’s overall scientific strategy, as well as oversee research operations to support the success of the Institute’s scientific initiatives.
We’ve learned a lot from lymphocytic choriomeningitis virus—now the time has come to fight it
Key Findings: There are no vaccines or therapies available for lymphocytic choriomeningitis virus (LCMV) infection. This pathogen spreads easily and
Awards & Honors
- 2005-2009 Pew Scholar in the Biomedical Sciences
- 2012 American Association of Immunologists (AAI) BD Biosciences Investigator Award for outstanding, early-career research contributions to the field of Immunology
- 2016 Thompson Reuters ISI, Web of Science, Highly Cited Researcher. Ostensibly a tabulation of the most influential ~125 immunologists in the world during the period 2004-2014, based on high impact papers.
- 2017 Thompson Reuters ISI, Web of Science, Highly Cited Researcher.
- 2018 UCSF Immunology Postdoctoral Fellows invited speaker.
- 2018 Stanford University Immunology Graduate Students invited speaker.
- 2018 University of Chicago Committee on Immunology (COI) Graduate Student body invited speaker.
- 2018 Thompson Reuters ISI, Web of Science, Highly Cited Researcher.
- 2019 Best of Immunity 2018 (Abbott et al. Immunity 2018)
- 2019 Cancer Research Institute’s (CRI) Fredrick W. Alt Award for New Discoveries in Immunology
- 2019 Web of Science, Highly Cited Researcher. Clarivate. Top 0.1% of researchers during the period 2007-2017 based on high impact papers published during that period.
- 2019 Elected Fellow. American Association for the Advancement of Science (AAAS).
- 2020 Web of Science, Highly Cited Researcher. Clarivate.
- 2020 Altmetric. #1 public attention ranked paper ever in Cell. Grifoni et al., 2020
- 2020 Altmetric. #1 public attention ranked paper ever in Nature Reviews Immunology. Sette and Crotty 2020
- 2020 Nature top 10 COVID papers of 2020. Dan et al. COVID-19 immune memory.
- 2021 NIH MERIT Award
- 2021 University of Wisconsin, Cellular and Molecular Pathology Student Choice Seminar














