
Overview
Our laboratory focuses on the involvement of myeloid cells in cardiovascular disease, cancer, and the long-term vascular consequences of COVID-19. Specifically, we study monocytes and neutrophils, as these are white blood cells that rapidly respond to viral infections, tumor cells, and early vascular changes in cardiovascular disease. Recent work by our laboratory has shown that a distinct subclass of monocytes termed “nonclassical monocytes” fight tumor metastasis. These nonclassical monocytes also prevent long-term vascular perturbations in recovered COVID-19 patients. We use high-dimensional methods such as CyTOF mass cytometry and single-cell RNA-sequencing to characterize these processes and identify novel myeloid cell types that may be important in treating or preventing disease.
Featured publications
Human monocyte heterogeneity as revealed by high-dimensional mass cytometry
Patrolling monocytes control tumor metastasis to the lung
Lab Members
 CH
CH
Catherine Hedrick
Professor Center for Autoimmunity and Inflammation, Center for Cancer Immunotherapy, Center for Sex-based Differences in the Immune SystemResearch Projects
CD8 T Cell Functions in Atherosclerosis
Epigenetic Regulation of Neutrophil and Monocyte Development in Disease
Immunoprofiling of Human Immune Cells in Cardiovascular Disease and Cancer
Regulation of Treg Function in Cardiovascular Disease
From the lab
Lynn Hedrick receives more than $500,000 for COVID-19 research
LA JOLLA—COVID-19 patients around the world are experiencing lasting cardiovascular issues, even after they’ve recovered from other symptoms. In fact,
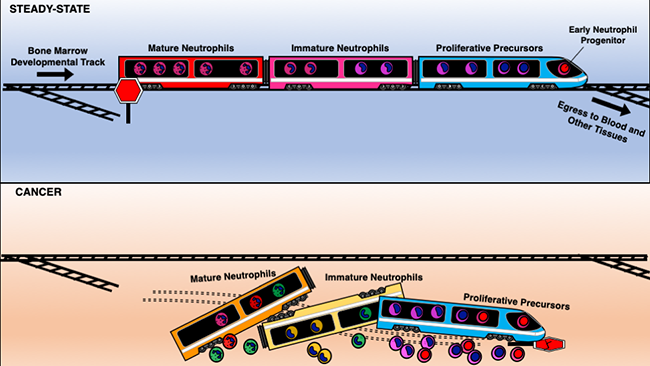
LJI team gets first-ever look at a rare but vital stem cell in humans
LA JOLLA—Neutrophils are the warriors of the immune system. They are always ready to spring to action to help heal
Immunoprofiling
The composition of an immune system varies not just from person to person, but over time—shifting as our health changes,
Awards & Honors
- Chair, GRC on Atherosclerosis, 2019
- Mentor of Women Award, ATVB Council, American Heart Association, 2015
- Jeffrey M. Hoeg Arteriosclerosis Thrombosis and Vascular Biology Award for Basic Science and Clinical Research, American Heart Association, 2014
- ATVB Vascular Biology Special Recognition Award, American Heart Association, 2013
- Co-Chair of 2012 Keystone Conference on Inflammation and Atherosclerosis
- Associate Editor, Arteriosclerosis Thrombosis and Vascular Biology, 2010
- Harrison Distinguished Chair in Molecular Physiology, University of Virginia, 2009
- Mary Jane Kugel Award, JDRF, 2005, 2008
- Recipient, Atorvastatin National Research Award for Cardiovascular Research, Pfizer, 2000
- Laverna Titus Young Investigator Award for Basic Science, American Heart Association, Los Angeles Affiliate, 1993, 1995
- Finalist, Young Investigator Award, International Atherosclerosis Society, 1994

